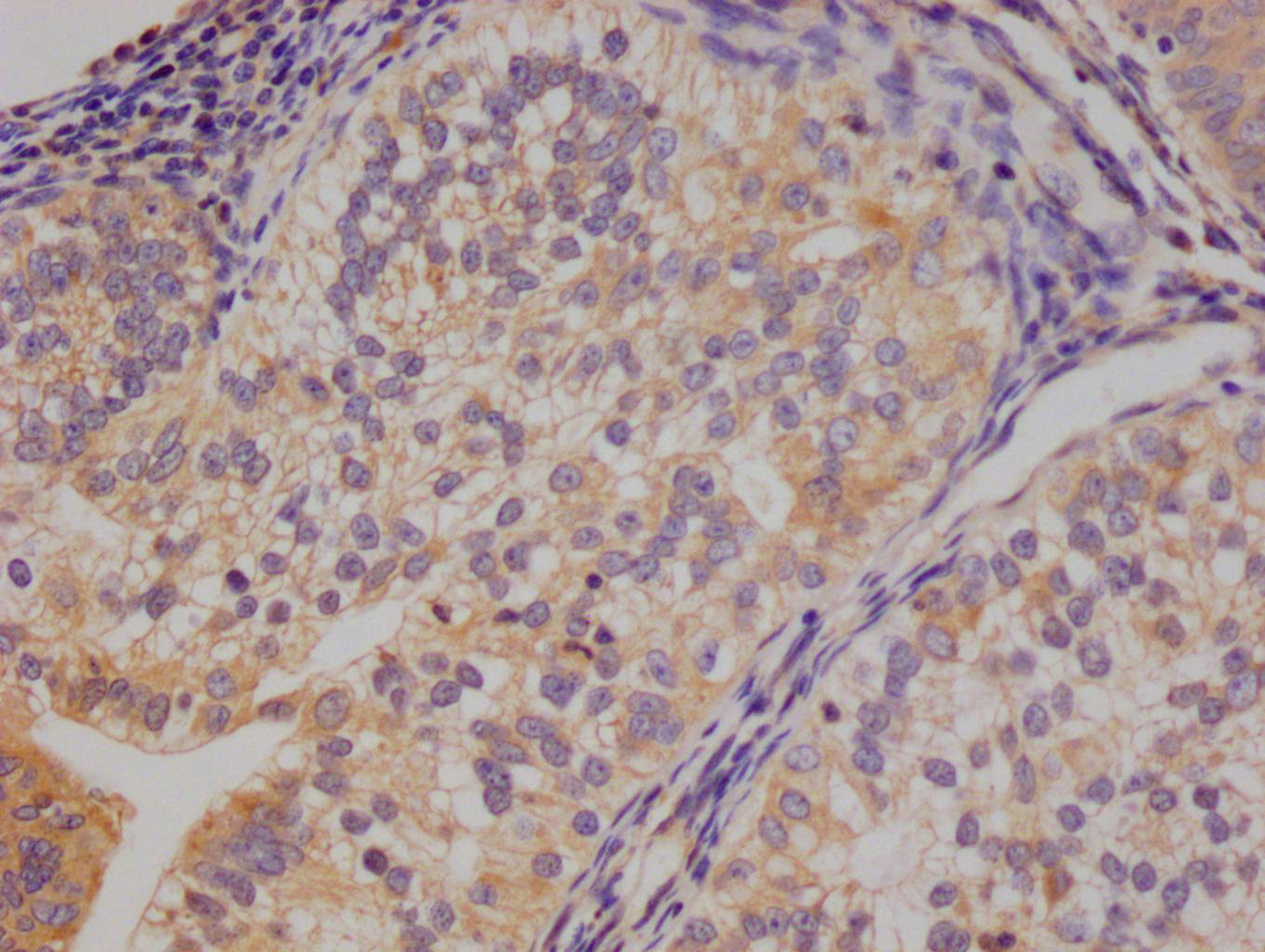
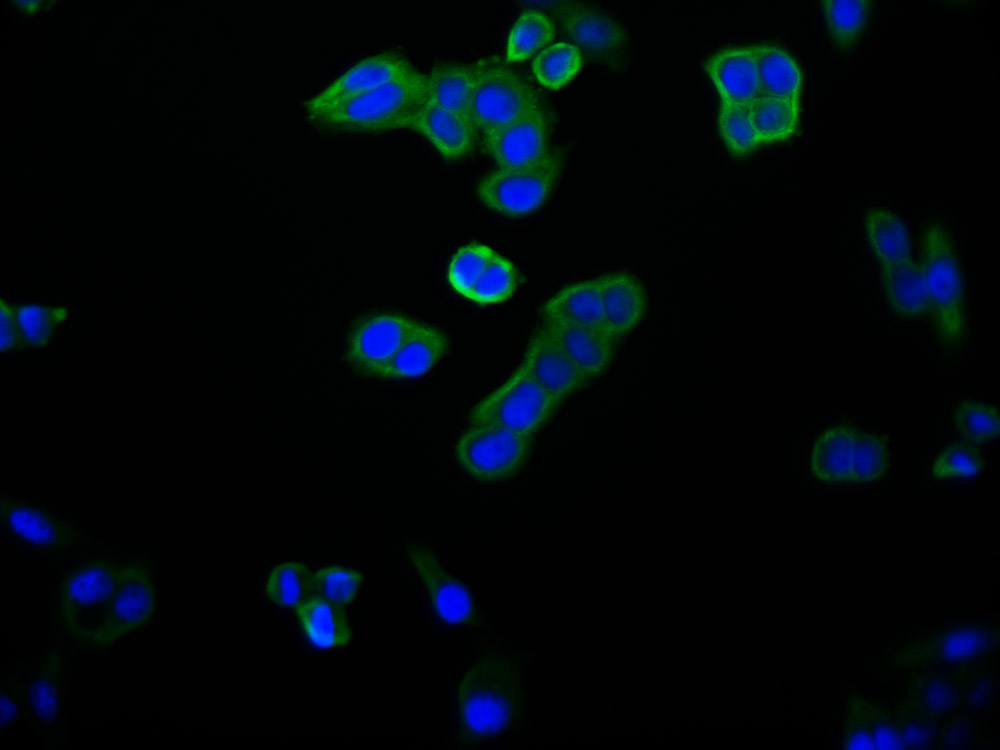
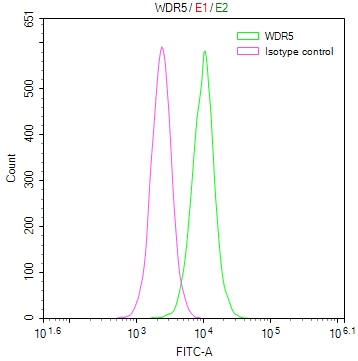
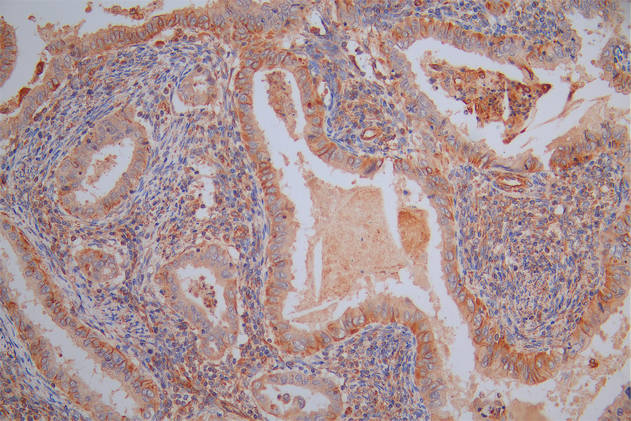
SIRT1 Antibody, Biotin conjugated
-
中文名稱:SIRT1兔多克隆抗體, Biotin偶聯(lián)
-
貨號:CSB-PA822202HD01HU
-
規(guī)格:¥880
-
其他:
產(chǎn)品詳情
-
產(chǎn)品名稱:Rabbit anti-Homo sapiens (Human) SIRT1 Polyclonal antibody
-
Uniprot No.:
-
基因名:
-
別名:75SirT1 antibody; hSIR2 antibody; hSIRT1 antibody; HST2 antibody; HST2, S. cerevisiae, homolog of antibody; NAD dependent deacetylase sirtuin 1 antibody; NAD dependent protein deacetylase sirtuin 1 antibody; NAD-dependent deacetylase sirtuin-1 antibody; OTTHUMP00000198111 antibody; OTTHUMP00000198112 antibody; Regulatory protein SIR2 homolog 1 antibody; SIR1_HUMAN antibody; SIR2 antibody; SIR2 like 1 antibody; SIR2 like protein 1 antibody; SIR2, S.cerevisiae, homolog-like 1 antibody; SIR2-like protein 1 antibody; SIR2ALPHA antibody; SIR2L1 antibody; Sirt1 antibody; SirtT1 75 kDa fragment antibody; Sirtuin (silent mating type information regulation 2 homolog) 1 (S. cerevisiae) antibody; Sirtuin 1 antibody; Sirtuin type 1 antibody
-
宿主:Rabbit
-
反應(yīng)種屬:Human
-
免疫原:Recombinant Human NAD-dependent protein deacetylase sirtuin-1 protein (2-747AA)
-
免疫原種屬:Homo sapiens (Human)
-
標記方式:Biotin
-
克隆類型:Polyclonal
-
抗體亞型:IgG
-
純化方式:>95%, Protein G purified
-
濃度:It differs from different batches. Please contact us to confirm it.
-
保存緩沖液:Preservative: 0.03% Proclin 300
Constituents: 50% Glycerol, 0.01M PBS, PH 7.4 -
產(chǎn)品提供形式:Liquid
-
應(yīng)用范圍:ELISA
-
Protocols:
-
儲存條件:Upon receipt, store at -20°C or -80°C. Avoid repeated freeze.
-
貨期:Basically, we can dispatch the products out in 1-3 working days after receiving your orders. Delivery time maybe differs from different purchasing way or location, please kindly consult your local distributors for specific delivery time.
相關(guān)產(chǎn)品
靶點詳情
-
功能:NAD-dependent protein deacetylase that links transcriptional regulation directly to intracellular energetics and participates in the coordination of several separated cellular functions such as cell cycle, response to DNA damage, metabolism, apoptosis and autophagy. Can modulate chromatin function through deacetylation of histones and can promote alterations in the methylation of histones and DNA, leading to transcriptional repression. Deacetylates a broad range of transcription factors and coregulators, thereby regulating target gene expression positively and negatively. Serves as a sensor of the cytosolic ratio of NAD(+)/NADH which is altered by glucose deprivation and metabolic changes associated with caloric restriction. Is essential in skeletal muscle cell differentiation and in response to low nutrients mediates the inhibitory effect on skeletal myoblast differentiation which also involves 5'-AMP-activated protein kinase (AMPK) and nicotinamide phosphoribosyltransferase (NAMPT). Component of the eNoSC (energy-dependent nucleolar silencing) complex, a complex that mediates silencing of rDNA in response to intracellular energy status and acts by recruiting histone-modifying enzymes. The eNoSC complex is able to sense the energy status of cell: upon glucose starvation, elevation of NAD(+)/NADP(+) ratio activates SIRT1, leading to histone H3 deacetylation followed by dimethylation of H3 at 'Lys-9' (H3K9me2) by SUV39H1 and the formation of silent chromatin in the rDNA locus. Deacetylates 'Lys-266' of SUV39H1, leading to its activation. Inhibits skeletal muscle differentiation by deacetylating PCAF and MYOD1. Deacetylates H2A and 'Lys-26' of H1-4. Deacetylates 'Lys-16' of histone H4 (in vitro). Involved in NR0B2/SHP corepression function through chromatin remodeling: Recruited to LRH1 target gene promoters by NR0B2/SHP thereby stimulating histone H3 and H4 deacetylation leading to transcriptional repression. Proposed to contribute to genomic integrity via positive regulation of telomere length; however, reports on localization to pericentromeric heterochromatin are conflicting. Proposed to play a role in constitutive heterochromatin (CH) formation and/or maintenance through regulation of the available pool of nuclear SUV39H1. Upon oxidative/metabolic stress decreases SUV39H1 degradation by inhibiting SUV39H1 polyubiquitination by MDM2. This increase in SUV39H1 levels enhances SUV39H1 turnover in CH, which in turn seems to accelerate renewal of the heterochromatin which correlates with greater genomic integrity during stress response. Deacetylates 'Lys-382' of p53/TP53 and impairs its ability to induce transcription-dependent proapoptotic program and modulate cell senescence. Deacetylates TAF1B and thereby represses rDNA transcription by the RNA polymerase I. Deacetylates MYC, promotes the association of MYC with MAX and decreases MYC stability leading to compromised transformational capability. Deacetylates FOXO3 in response to oxidative stress thereby increasing its ability to induce cell cycle arrest and resistance to oxidative stress but inhibiting FOXO3-mediated induction of apoptosis transcriptional activity; also leading to FOXO3 ubiquitination and protesomal degradation. Appears to have a similar effect on MLLT7/FOXO4 in regulation of transcriptional activity and apoptosis. Deacetylates DNMT1; thereby impairs DNMT1 methyltransferase-independent transcription repressor activity, modulates DNMT1 cell cycle regulatory function and DNMT1-mediated gene silencing. Deacetylates RELA/NF-kappa-B p65 thereby inhibiting its transactivating potential and augments apoptosis in response to TNF-alpha. Deacetylates HIF1A, KAT5/TIP60, RB1 and HIC1. Deacetylates FOXO1 resulting in its nuclear retention and enhancement of its transcriptional activity leading to increased gluconeogenesis in liver. Inhibits E2F1 transcriptional activity and apoptotic function, possibly by deacetylation. Involved in HES1- and HEY2-mediated transcriptional repression. In cooperation with MYCN seems to be involved in transcriptional repression of DUSP6/MAPK3 leading to MYCN stabilization by phosphorylation at 'Ser-62'. Deacetylates MEF2D. Required for antagonist-mediated transcription suppression of AR-dependent genes which may be linked to local deacetylation of histone H3. Represses HNF1A-mediated transcription. Required for the repression of ESRRG by CREBZF. Deacetylates NR1H3 and NR1H2 and deacetylation of NR1H3 at 'Lys-434' positively regulates transcription of NR1H3:RXR target genes, promotes NR1H3 proteosomal degradation and results in cholesterol efflux; a promoter clearing mechanism after reach round of transcription is proposed. Involved in lipid metabolism. Implicated in regulation of adipogenesis and fat mobilization in white adipocytes by repression of PPARG which probably involves association with NCOR1 and SMRT/NCOR2. Deacetylates p300/EP300 and PRMT1. Deacetylates ACSS2 leading to its activation, and HMGCS1 deacetylation. Involved in liver and muscle metabolism. Through deacetylation and activation of PPARGC1A is required to activate fatty acid oxidation in skeletal muscle under low-glucose conditions and is involved in glucose homeostasis. Involved in regulation of PPARA and fatty acid beta-oxidation in liver. Involved in positive regulation of insulin secretion in pancreatic beta cells in response to glucose; the function seems to imply transcriptional repression of UCP2. Proposed to deacetylate IRS2 thereby facilitating its insulin-induced tyrosine phosphorylation. Deacetylates SREBF1 isoform SREBP-1C thereby decreasing its stability and transactivation in lipogenic gene expression. Involved in DNA damage response by repressing genes which are involved in DNA repair, such as XPC and TP73, deacetylating XRCC6/Ku70, and facilitating recruitment of additional factors to sites of damaged DNA, such as SIRT1-deacetylated NBN can recruit ATM to initiate DNA repair and SIRT1-deacetylated XPA interacts with RPA2. Also involved in DNA repair of DNA double-strand breaks by homologous recombination and specifically single-strand annealing independently of XRCC6/Ku70 and NBN. Transcriptional suppression of XPC probably involves an E2F4:RBL2 suppressor complex and protein kinase B (AKT) signaling. Transcriptional suppression of TP73 probably involves E2F4 and PCAF. Deacetylates WRN thereby regulating its helicase and exonuclease activities and regulates WRN nuclear translocation in response to DNA damage. Deacetylates APEX1 at 'Lys-6' and 'Lys-7' and stimulates cellular AP endonuclease activity by promoting the association of APEX1 to XRCC1. Increases p53/TP53-mediated transcription-independent apoptosis by blocking nuclear translocation of cytoplasmic p53/TP53 and probably redirecting it to mitochondria. Deacetylates XRCC6/Ku70 at 'Lys-539' and 'Lys-542' causing it to sequester BAX away from mitochondria thereby inhibiting stress-induced apoptosis. Is involved in autophagy, presumably by deacetylating ATG5, ATG7 and MAP1LC3B/ATG8. Deacetylates AKT1 which leads to enhanced binding of AKT1 and PDK1 to PIP3 and promotes their activation. Proposed to play role in regulation of STK11/LBK1-dependent AMPK signaling pathways implicated in cellular senescence which seems to involve the regulation of the acetylation status of STK11/LBK1. Can deacetylate STK11/LBK1 and thereby increase its activity, cytoplasmic localization and association with STRAD; however, the relevance of such activity in normal cells is unclear. In endothelial cells is shown to inhibit STK11/LBK1 activity and to promote its degradation. Deacetylates SMAD7 at 'Lys-64' and 'Lys-70' thereby promoting its degradation. Deacetylates CIITA and augments its MHC class II transactivation and contributes to its stability. Deacetylates MECOM/EVI1. Deacetylates PML at 'Lys-487' and this deacetylation promotes PML control of PER2 nuclear localization. During the neurogenic transition, represses selective NOTCH1-target genes through histone deacetylation in a BCL6-dependent manner and leading to neuronal differentiation. Regulates the circadian expression of several core clock genes, including ARNTL/BMAL1, RORC, PER2 and CRY1 and plays a critical role in maintaining a controlled rhythmicity in histone acetylation, thereby contributing to circadian chromatin remodeling. Deacetylates ARNTL/BMAL1 and histones at the circadian gene promoters in order to facilitate repression by inhibitory components of the circadian oscillator. Deacetylates PER2, facilitating its ubiquitination and degradation by the proteosome. Protects cardiomyocytes against palmitate-induced apoptosis. Deacetylates XBP1 isoform 2; deacetylation decreases protein stability of XBP1 isoform 2 and inhibits its transcriptional activity. Deacetylates PCK1 and directs its activity toward phosphoenolpyruvate production promoting gluconeogenesis. Involved in the CCAR2-mediated regulation of PCK1 and NR1D1. Deacetylates CTNB1 at 'Lys-49'. In POMC (pro-opiomelanocortin) neurons, required for leptin-induced activation of PI3K signaling. In addition to protein deacetylase activity, also acts as protein-lysine deacylase: acts as a protein depropionylase by mediating depropionylation of Osterix (SP7). Deacetylates SOX9; promoting SOX9 nuclear localization and transactivation activity. Involved in the regulation of centrosome duplication. Deacetylates CENATAC in G1 phase, allowing for SASS6 accumulation on the centrosome and subsequent procentriole assembly. {ECO:0000250|UniProtKB:Q923E4, ECO:0000269|PubMed:11672523, ECO:0000269|PubMed:12006491, ECO:0000269|PubMed:12535671, ECO:0000269|PubMed:14976264, ECO:0000269|PubMed:14980222, ECO:0000269|PubMed:15126506, ECO:0000269|PubMed:15152190, ECO:0000269|PubMed:15205477, ECO:0000269|PubMed:15469825, ECO:0000269|PubMed:15692560, ECO:0000269|PubMed:16079181, ECO:0000269|PubMed:16166628, ECO:0000269|PubMed:16892051, ECO:0000269|PubMed:16998810, ECO:0000269|PubMed:17283066, ECO:0000269|PubMed:17290224, ECO:0000269|PubMed:17334224, ECO:0000269|PubMed:17505061, ECO:0000269|PubMed:17612497, ECO:0000269|PubMed:17620057, ECO:0000269|PubMed:17936707, ECO:0000269|PubMed:18203716, ECO:0000269|PubMed:18296641, ECO:0000269|PubMed:18485871, ECO:0000269|PubMed:18662546, ECO:0000269|PubMed:18687677, ECO:0000269|PubMed:19188449, ECO:0000269|PubMed:19220062, ECO:0000269|PubMed:19364925, ECO:0000269|PubMed:19690166, ECO:0000269|PubMed:19934257, ECO:0000269|PubMed:20097625, ECO:0000269|PubMed:20100829, ECO:0000269|PubMed:20203304, ECO:0000269|PubMed:20375098, ECO:0000269|PubMed:20620956, ECO:0000269|PubMed:20670893, ECO:0000269|PubMed:20817729, ECO:0000269|PubMed:20955178, ECO:0000269|PubMed:21149730, ECO:0000269|PubMed:21245319, ECO:0000269|PubMed:21471201, ECO:0000269|PubMed:21504832, ECO:0000269|PubMed:21555002, ECO:0000269|PubMed:21698133, ECO:0000269|PubMed:21701047, ECO:0000269|PubMed:21775285, ECO:0000269|PubMed:21807113, ECO:0000269|PubMed:21841822, ECO:0000269|PubMed:21890893, ECO:0000269|PubMed:21947282, ECO:0000269|PubMed:22274616, ECO:0000269|PubMed:24415752, ECO:0000269|PubMed:24824780, ECO:0000269|PubMed:30193097, ECO:0000269|PubMed:31722219}.; [Isoform 2]: Deacetylates 'Lys-382' of p53/TP53, however with lower activity than isoform 1. In combination, the two isoforms exert an additive effect. Isoform 2 regulates p53/TP53 expression and cellular stress response and is in turn repressed by p53/TP53 presenting a SIRT1 isoform-dependent auto-regulatory loop. {ECO:0000269|PubMed:20975832}.; (Microbial infection) In case of HIV-1 infection, interacts with and deacetylates the viral Tat protein. The viral Tat protein inhibits SIRT1 deacetylation activity toward RELA/NF-kappa-B p65, thereby potentiates its transcriptional activity and SIRT1 is proposed to contribute to T-cell hyperactivation during infection. {ECO:0000269|PubMed:18329615}.; [SirtT1 75 kDa fragment]: Catalytically inactive 75SirT1 may be involved in regulation of apoptosis. May be involved in protecting chondrocytes from apoptotic death by associating with cytochrome C and interfering with apoptosome assembly. {ECO:0000269|PubMed:21987377}.
-
基因功能參考文獻:
- These results suggest that resveratrol induces chondrosarcoma cell apoptosis via a SIRT1-activated NF-kappaB (p65 subunit of NF-kappaB complex)deacetylation and exhibits anti-chondrosarcoma activity in vivo. PMID: 28600541
- I157172 induced upregulation of SIRT1, and downregulation of acetyl-STAT3. PMID: 30365149
- SIRT1-mediated H3K9 deacetylation helps to maintain gene repression but is not required for the direct ZEB2 repressive function.SIRT1 activity maintains the stability of ZEB2-induced RAB25 repression. PMID: 30445998
- Data show that long non-coding RNA MALAT1 (MALAT1) repressed sirtuin 1 (SIRT1) expression through targeting forkhead box protein O1 (Foxo1). PMID: 29928873
- SIRT1 had a pivotally protective role in the regulation of ADSCs aging and apoptosis induced by H2O2 PMID: 29803744
- plasma levels correlated inversely with all studied adiposity and atherogenicity indices in metabolic syndrome patients with and without prediabetes PMID: 29779969
- Increased SIRT1 activity protects against diabetes-induced podocyte injury and effectively mitigates the progression of diabetic kidney disease. PMID: 29477240
- the Sirt1 carboxyl-domain is an ATP-repressible domain that is transferrable to other proteins PMID: 28504272
- Adipose tissue sirtuin 1 was related to insulin sensitivity. The relationship was still present after controlling for BMI, however, it disappeared after controlling for adipose tissue SLC2A4. Muscle sirtuin 1 was not related to insulin sensitivity. PMID: 29417372
- data suggest that SIRT1 is an oncogenic factor in breast cancer cells and can be involved in the progression of breast cancer by inhibiting p53 and activating POLD1 PMID: 29807012
- SIRT1 expression is significantly upregulated in paclitaxel-resistant cervical cancer tissues and cell lines compared to normal tissues or PTX-sensitive CC tissues and cell lines. Knockdown of SIRT1 inhibited the cell proliferation, promoted cell cycle arrest and apoptosis of PTX-sensitive CC cells, and decreased the expression of MDR proteins. PMID: 29808798
- In our retrospective study, high SIRT1 expression significantly correlated with vascular invasion and a worse prognosis in colorectal cancer PMID: 30082156
- SIRT1 polymorphisms and their expression were associated with the presence of alcoholic fatty liver disease (AFLD), and there was a close relationship among four SNPs and body mass index in AFLD patients, but no SNP was related to its expression. PMID: 29189974
- The variable role of SIRT1 in the maintenance and differentiation of mesenchymal stem cells. PMID: 29715067
- Results indicate that SIRT1 may promote the metastasis of chondrosarcoma by inducing epithelial-mesenchymal transition and can be a potential molecular target for chondrosarcoma therapy. PMID: 28112277
- Regulation of transmembrane-4-L-six-family-1 (TM4SF1) on bladder cancer cell could be induced by peroxisome proliferator-activated receptor gamma (PPARgamma)-sirtuin 1 (SIRT1) feedback loop. PMID: 29175458
- Results present evidence that SIRT1 plays an essential role in regulating the transcription of CLDN5 likely by modifying and modulating the activity of KLF4 in ovarian cancer cells. PMID: 28888043
- results suggest that seminal SIRT1 expression has a role in male infertility PMID: 29359516
- SIRT1 may promote the transformation of tumor cells by inducing the epithelial-mesenchymal transition. PMID: 29656187
- relevance of the discovered Sirt1-Smad2 interaction for the regulation of TGFbeta-dependent gene transcription PMID: 29187201
- Bioinformatics and the dual luciferase reporter assay analysis results demonstrated that miR-29a specifically targeted the 3'-UTR of SIRT1 mRNA and regulated its protein expression. Increased SIRT1 expression rescued the inhibited effects induced by miR-29a overexpression in HCC cells. PMID: 29630527
- High Sirt1 Expression is associated with Gastric cancer. PMID: 29693338
- Sirt1 protects against oxidative stress-induced apoptosis in fibroblasts from psoriatic patients. PMID: 29799444
- Investigated the effect of statins on the expression of sirtuin 1 (SIRT1) and endothelial nitric oxide synthase 3 (eNOS) proteins in young premature myocardial infarction (PMI) patients. Found patients with PMI who were taking statins had a markedly higher level of SIRT1 compared with the controls. The level of eNOS protein was considerably lower in PMI patients compared with the control group. PMID: 29664427
- Clinical data are the first to identify SIRT1 as an important regulator of hepatocellular function in human liver transplants under ischemia/reperfusion stress. PMID: 28719070
- suggests SIRT1 may serve as a predictor of poor prognosis in esophageal squamous cell carcinoma, and its mediated tumor-promoting role might be associated with the overexpression of EGFR protein in esophageal squamous cell carcinoma PMID: 29625788
- findings demonstrate a new mechanism for the activation of SIRT1 under stress conditions and suggest a novel potential therapeutic target for preventing age-related diseases and extending healthspan PMID: 29133780
- This study represents the important role of Sirt1 and senescence in the regulation of beige adipocyte differentiation during aging. PMID: 29678576
- Baicalin activated the SIRT1/AMPK and mechanistic target of rapamycin (mTOR), and SIRT1/AMPK and matrix metalloproteinase (MMP) signaling in A549 and H1299 cells in a dose-dependent manner. siRNA silencing of SIRT1 and AMPK reduced the effects of baicalin on cell proliferation and migration. PMID: 29632297
- We provide a comprehensive overview of recent developments on the molecular signaling pathways controlled by SIRT1 and SIRT6, two post-translational modifiers proven to be valuable tools to dampen inflammation and oxidative stress at the cardiovascular levell PMID: 28661724
- SIRT1 was identified as a direct target of miR-212 and its expression was inversely correlated with miR-212 expression in thyroid cancer tissues. Overexpression of SIRT1 could effectively rescue miR-212 mimic-induced suppression of cell proliferation, migration and invasion in TPC-1 thyroid cancer cell line. PMID: 29207181
- The present study is the first to report a significant association between SIRT1 polymorphisms and antisocial personality in adolescents. PMID: 28439078
- Studied effects of dexamethasone on gene expression regulation of sirtuin 1 (SIRT1), interleukin 6 (IL6), and endothelin 1 (EDN1) in gingival derived aging stem cells. Dexamethasone downregulated expression of SIRT1 and IL6 but upregulated EDN1 in stem cells. PMID: 29302812
- miR-200a and its target gene, SIRT1, may exert a possible role in induction of apoptosis in dopaminergic neurons. PMID: 29936262
- The rhuschalcone I analogue (9) showed the best activity against sirt1, with an IC50 value of 40.8 microM. Based on the docking experiments, suggestions for improving the biological activities of the newly identified hit compounds have been provided. PMID: 29443909
- Results indicated that SIRT1 contributes to the neuroprotection of salidroside against MPP(+) -induced apoptosis and oxidative stress, in part through suppressing of mitogen-activated protein kinase (MAPK) pathways. PMID: 28851138
- decreased SIRT1 expression and its SUMOylation by SUMO1 and SUMO2/3 may be associated with the development of bronchopulmonary dysplasia. PMID: 29115559
- The nicotinamide adenine dinucleotide (NAD)-dependent deacetylase SIRT1 acts as an energy sensor and negatively regulates poly(A)RNA transport via deacetylating a poly(A)-binding protein, PABP1. PMID: 28756945
- L-Carnitine alleviated epithelial mesenchymal transformation-associated renal fibrosis caused by perfluorooctanesulfonate through a Sirt1- and PPARgamma-dependent mechanism. PMID: 28973641
- High SIRT1 expression is associated with hepatocellular carcinoma. PMID: 28677784
- No significant association has been discovered between SIRT1 polymorphisms and diabetic foot severity or characteristics PMID: 29995800
- A significant correlation between the physical activity level and peripheral blood mononuclear cell SIRT1 and FOXO1 mRNA expression was found in COPD patients. PMID: 29138552
- Defective sirtuin-1 was found to increase IL-4 expression through acetylation of GATA-3 in patients with severe asthma compared with healthy controls. PMID: 26627546
- SIRT1 gene polymorphisms can have direct and indirect effects on the pathogenesis of coronary artery diseases. PMID: 29885463
- miR-146 exerted protective functions might be via up-regulation of Sirt1 thereby blocking NF-kappaB and Notch pathways. PMID: 29229881
- results suggested that SIRT1 deficiency in Bladder cancer cells could suppress cell viability by activating antioxidant response and inducing cell cycle arrest possibly via FOXO3a-related pathways. PMID: 29147649
- These observations provide further evidence for a critical tumor-suppressive role of the miR-200a in renal cell carcinoma (RCC) in addition to identifying a novel regulatory mechanism, which may contribute to SIRT1 upregulation in RCC. PMID: 28717923
- Results showed that RSV or overexpression of SIRT1 elicited inhibitory effects on NMDA-induced excitotoxicity including a decrease in cell viability, an increase in lactate dehydrogenase (LDH) release, and a decrease in the number of living cells as measured by CCK-8 assay, LDH test, and Calcein-AM and PI double staining. PMID: 29081884
- results indicate that FOXO1 inhibits gastric cancer (GC) growth and angiogenesis under hypoxic conditions via inactivation of the HIF-1alpha-VEGF pathway, possibly in association with SIRT1; thus, development of treatment modalities aiming at this pathway might be useful for treating GC PMID: 25761483
- Data suggest the activity restoration role of resveratrol toward some "loose-binding" substrates of sirtuin 1 (SIRT1), and has implications for the rational design of new substrate-specific SIRT1 modulators. PMID: 27901083
顯示更多
收起更多
-
亞細胞定位:Nucleus, PML body. Cytoplasm. Nucleus.; [SirtT1 75 kDa fragment]: Cytoplasm. Mitochondrion.
-
蛋白家族:Sirtuin family, Class I subfamily
-
組織特異性:Widely expressed.
-
數(shù)據(jù)庫鏈接:
Most popular with customers
-
-
YWHAB Recombinant Monoclonal Antibody
Applications: ELISA, WB, IF, FC
Species Reactivity: Human, Mouse, Rat
-
-
-
-
-
-